
Sponsors often have rigid timing for study milestones, and the clock starts as soon as the CRO contract is signed. 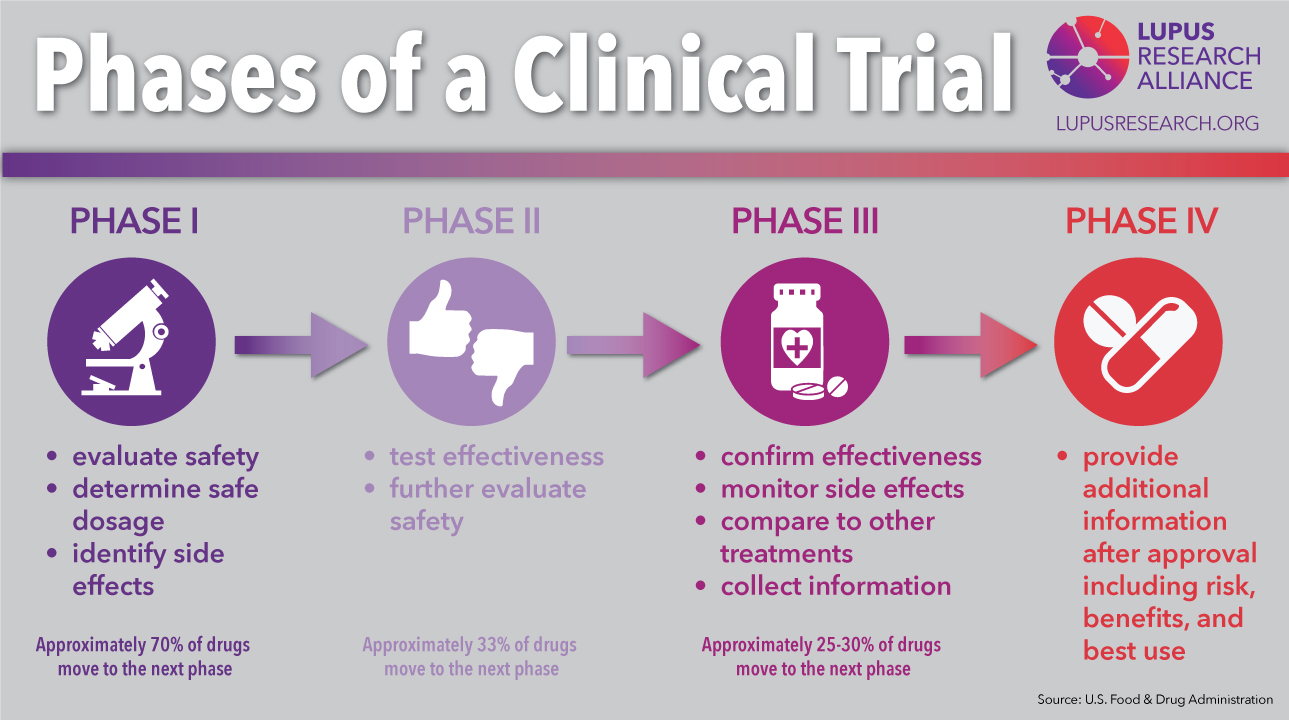
Here are some other start-up recommendations we offer to current and prospective customers: Having an itinerant launch crew that moves from trial to trial guarantees that every study gets our best start-up expertise. All these project launch managers do is study start-up, and here’s why: Clinical trials typically last for years, and team members who stay aboard a trial from start to finish lack the opportunity to develop the highly specific skills and disciplines that ensure an effective launch. Once the study is awarded, it becomes the responsibility of a dedicated Project Launch Manager and a focused start-up team whose sole focus is an effective and efficient start-up, laying the foundation for successful execution. It’s a lot of effort on our investment dime, but the payoff can be considerable in a smoother study launch without the false starts and missteps that often result from the absence of careful planning. Assessing large amounts of data at the pre-award stage is vital to putting the study on the right path and is indispensable in developing the contractual agreement post-award. There’s no better time to hone our understanding of what’s important to the sponsor: first site initiated, first patient in, or whichever other goal is most relevant. The process actually starts before the study is even awarded, at the RFP stage. That way, our sponsors can focus more on science and less on … everything else. The underlying goal is to simplify by doing a lot of advance work and by giving sponsors a single CRO interface who takes on all the worries - from protocol design to regulatory affairs, from site selection to ethics. That’s why we created a department that consolidates all the expertise needed to get trials off on the right foot. 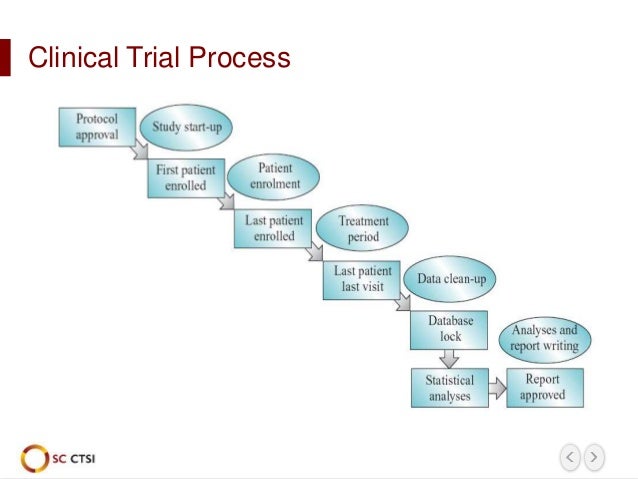
The waste of money and time - commodities that are chronically scarce among the biotech and specialty pharma companies that comprise most of our customer base - can be devastating. Botch the start-up and you’ll expend great effort correcting course and playing catch-up. You get one chance to initiate a clinical trial.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
